The aim of this article is to understand the following
In 1866 Langdon Down noted that common characteristics of patients with trisomy 21 are skin deficient in elasticity, giving the appearance of being too large for the body, and flat face with a small nose. In the 1990s, it was realized that the excess skin of individuals with Down’s syndrome could be visualized by ultrasonography as increased nuchal translucency in the third month of intrauterine life.
The first method of screening for Down syndrome introduced in the 1970s was the use of maternal age. Any woman above the age of 35 was offered amniocentesis. A large number of women underwent amniocentesis and 70% of Down syndrome babies that were born to younger women were missed.
In the late 1980s, a new method of screening was introduced that took into account not only maternal age but also the concentration of various fetoplacental products in the maternal circulation (alphafetoprotein (AFP), unconjugated estriol (uE3), human chorionic gonadotropin (hCG). This was labeled as triple screen. Later inhibin-A was added and the quadruple screen came into vogue.
In the 90s, First Trimester Screen with the use of maternal age and Nuchal translucency at 11 – 13 + 6 weeks was introduced by the Fetal Medicine Foundation, London,UK.
Subsequently, maternal age was combined with fetal Sonography markers like NT, Nasal bone (NB), Ductus venosus Doppler (DV), Tricuspid Regurgitation (TR) and maternal serum biochemistry (free b-hCG and PAPP-A) in the first trimester. Use of multiple parameters orcombined testhelped to significantly increase detection rates.
Therefore, it is extremely important to emphasize the need to karyotype in any child with Down syndrome, even when the diagnosis is very obvious and clear, for proper counseling in future pregnancy.
In Normal human being , we have total 46 chromosome ( 23 pairs ). Last 23 pair is XX in case of female and XY in case of male.
Down syndrome is a chromosomal abnormality where the child is born with total 47 chromosome, an extra one chromosome in 21st pair so there is total 3 Pcs of chromosome 21.
Down syndrome occurs in about 1 in 800 pregnancies in India. Any woman can have a baby with Down syndrome. The risk varies with the
Mother’s age. As the maternal age increases chances of having Down syndrome increases. At the maternal age of 21 years, chance is 1:1200 approx., which increases to 1:250 at the age of 35 and 1:80 at the age of 40!
70% of babies with Down syndrome are born to younger mothers and therefore screening should be offered to all women.
Baseline risk and likelihood ratios
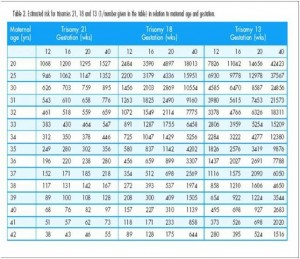
Every individual has a baseline risk of a problem occurring. This is also called a priori risk. With respect to Down syndrome, this risk varies for each woman based on her age. Hence in a given practice, it is important to consistently use the same table. The Fetal Medicine Foundation has published tables with maternal age related risk of trisomy 21 at different gestational ages. This table accounts for the natural selection of Down syndrome with advancing gestational age. It is mentioned below:
Maternal age related risk as per FMF.
Yes, because each of these cytogenetic abnormalities has got different risk of recurrence.
There are three types of cytogenetic abnormalities seen in Down syndrome.
Screening is a process that helps to identify people “at risk” for a condition or a problem in an asymptomatic and large population , to picked up one where there is some treatment or intervention possible.
Those who are found to be “at increased risk” of a condition may need another test to confirm if indeed the problem is present or not.
Identifying markers on ultrasound or biochemistry can do screening.
Typically, the presence of a marker alters the baseline or a priori risk.
Every individual has a baseline risk of a problem occurring. This is also called a priori risk. With respect to Down syndrome, this risk varies for each woman based on her age. Hence in a given practice, it is important to consistently use the same table. The Fetal Medicine Foundation has published tables with maternal age related risk of trisomy 21 at different gestationalages. This table accounts for the natural selection of Down syndrome with advancing gestational age.(mentioned before)
The priori risk is modified or changed by the result of the screening test during 12 weeks scan – NT with or with out NB and Free b-hCG& PAPP-A.
These screening tests e.g. ultrasonography markers or biochemical markers act as filters. Each filter has a likelihood ratio( its strength of association with Trisomy 21), which is the number of times by which the presence of the marker increases the baseline risk. If the likelihood ratio of a test is 5, then the chance of the baby having Down syndrome increases five times. The likelihood ratio of each test varies depending on whether it is a “good” filter or a “poor” filter.
If we are applying a series of tests, then the baseline or a priori risk multiplied by the likelihood ratio of one test gives a new risk. This risk then forms the baseline risk upon which the likelihood ratio of the second test is applied. This is called as sequential screening.
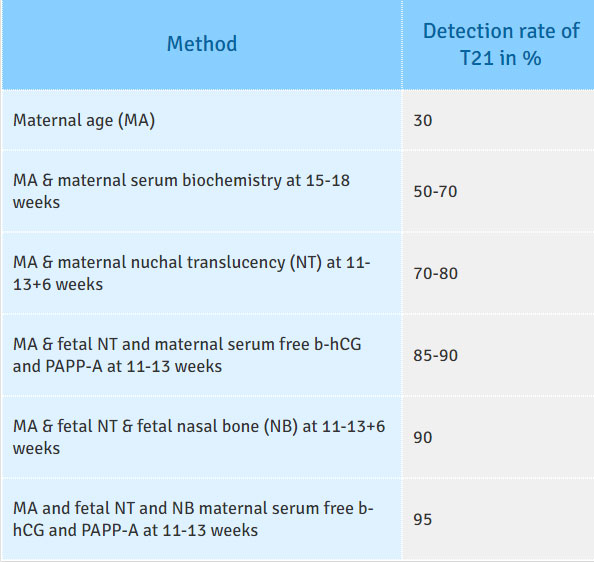
Table: Comparison of the detection rates (DR), for a false positive rate of 5%, of different methods of screening for trisomy 21.
he risk cut-off is an arbitrary number or line beyond which a decision is made to offer a further diagnostic test, which is a CVS or amniocentesis. This cut-off is kept in such a fashion that the invasive testing is offered to 5% of the highest risk population. The cut-off for a particular program is dictated by policy.
Currently in India, we are using a cut-off of 1:100.
As the efficacy of the screening test improves, the detection rate increases with a low false positive rate.
The optimal time for doing a first trimester screening (FTS) is from 11 weeks to 13 weeks 6 days. The CRL should be a minimum of 45 mm to a maximum of 84 mm.Prediction of risk from NT is best from 11 weeks onwards and NT regresses by 14 weeks.The biochemistry markers that are used for first trimester screening are free b-hCG and PAPP-A these are most sensitive between 9 – 13 weeks.
When a result of screening comes back as screen negative, no further testing is generally warranted because it suggests a ‘low risk’ of having a baby with Down syndrome. When a woman screens positive, it does not mean that the baby is affected. This only puts her in a higher risk group and further invasive testing is offered for fetal karyotyping.
The process of screening, with biochemistry, involves taking note of some maternal criteria. These include
The first scan in early pregnancy should be undertaken ideally between 11-13+6 weeks. The purpose of this scan is to establish:
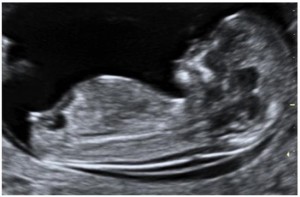
Accurate dating of pregnancy is the first step to the process of screening. This is established by a scan by taking an accurate CRL.
Asinterpretation of NT is based on CRL, it is important that the CRL is taken in the standard position.
figure: ideal CRL
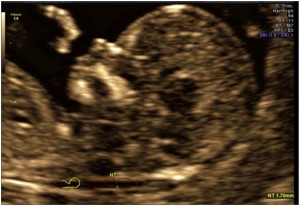
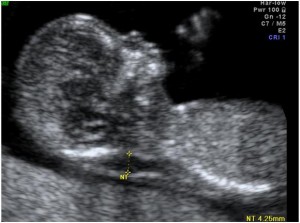
Nuchal translucency is the sonographic appearance of subcutaneous accumulation of fluid behind the fetal neck in the first trimester of pregnancy.
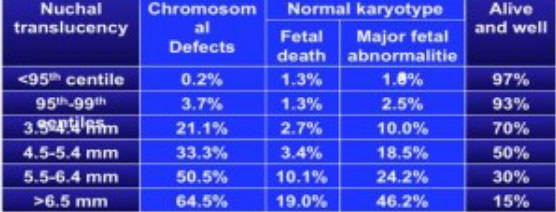
The incidence of chromosomal and other abnormalities is related to the size, rather than the appearance of NT.Fetal NT increases with gestation. Each NT value for a specific CRL has a likelihood ratio (LR) and larger the NT, higher the LR.
The Nuchal translucency measurement is to be taken according to the FMF specifications.
figure: NT

www.fetalmedicine.com
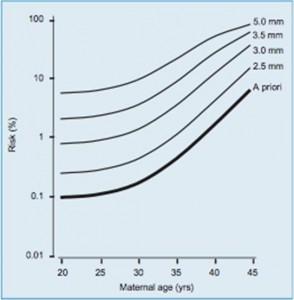
we can very well see from above chart that as that NT increases, chances of risk of chromosomal malformations increases.
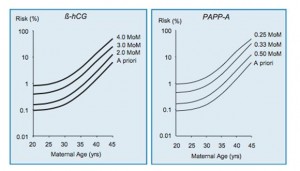
This involves a simple blood test which measures the free beta- hCG and PAPP-A. The level of free b-hCG in maternal blood normally decreases with gestation. The level of PAPP-A in maternal blood normally increases with gestation. In trisomy 21 pregnancies free b-hCG is increased, and PAPP-A level is decreased.
For a given gestation, each b-hCG and PAPP-A level represents a likelihood ratio that is multiplied by the priori risk to calculate the new risk. The higher the level of b-hCG and the lower the level of PAPP-A the higher the risk for trisomy 21.

At present, the cost effectiveness is questionable for the developing or poor countries like India.
As almost about 80 % of the fetuses are screened by NT (ultrasound markers e.g. Nasal bone, ductus venosus )and can be classified as LOW risk.
About 5% are classified as HIGH risk on the basis of NT (ultrasound markers e.g. Nasal bone, ductus venosus) , and offered invasive testing for either for NIPT or chromosomal study after CVS.
Rest of about 15% who are in to intermediate risk, we can take help of biochemical markers to put them in to HIGH or LOW risk. If still couple wants to confirm the diagnosis, couple may be offered the prenatal invasive diagnosis or NIPT.
The advantages of first-trimester screening extend beyond the observed higher detection rates. The main advantage for the majority of women is the earlier reassurance provided to those with ‘low-risk’ results.
Those who are screen positive can have a CVS and diagnosis by the 14th week. Complication rates are also lower at earlier gestations for termination of pregnancy in case of fetal chromosomal anomalies.
A range of abnormalities other than aneuploidy has been associated with increased nuchal translucency. These include cardiac anomalies, diaphragmatic hernia, skeletal dysplasia, and abnormal lymphatic drainage associated with neuromuscular disorders and many more.
Nicolaides KH et al 23 mentioned that almost 43.6% of fetal anomalies were detected at 11-13+6 weeks. The 11- 13+6 weeks scan detected all cases of acrania,
alobarholoprosencephaly,exomphalos, gastroschisis, megacystis and body stalk anomaly, 77% of absent hand or foot,50% of diaphragmatic hernia, 50% of lethal skeletal dysplasias, 60% of polydactyly, 34% ofmajor cardiac defects, 5% of facial clefts and 14% of open spina bifida, but none of agenesis ofthe corpus callosum, cerebellar or vermian hypoplasia, echogenic lung lesions, bowelobstruction, most renal defects or talipes. Nuchal translucency (NT) was above the 95thpercentile in 34% of fetuses with major cardiac defects.
Therefore, identification of an increased nuchal translucency measurement should prompt not only a diagnostic test for aneuploidy, but also a thorough anatomic survey for structural anomalies and a detailed fetal echocardiogram in the case of a normal karyotype.
A significant number of anomalies can be picked up at this gestational age. With the increasing use of higher resolution machines a detailed anatomical survey can be done at the 11- 13 week scan.
In our country the baseline awareness of Down syndrome is low and that for the screening test even less. Counseling needs to be one on one, ensuring that the information is understood correctly.
Once the screening is done, the result is again explained. If screen positive, the details of invasive testing are discussed and appointment for the same scheduled.
Important table to understand:

Check if the risk prediction has included the NT + other USG markers and +/- biochemistry and the combined risk has been predicted using ASTRAIA software provided by FMF or AstraiaGmBH , Germany.
The report should mention both the woman’s age related risk and give the estimated risk.
Training and certification is an essential part of setting up a screening service. The Fetal Medicine Foundation has made it simple to take an online course followed by practical training to obtain such certification. This information is available at www.fetalmedicine.com
During the last 30 years, extensive research has aimed at developing a non-invasive method for prenatal diagnosis based on the isolation and examination of fetal cells found in the maternal circulation. About 1 in 103–107 nucleated cells in maternal blood are fetal. The proportion of fetal cells can be enriched to about 1 in 10–100 by techniques such as magnetic cell sorting (MACS) or fluorescence activated cell sorting (FACS) after attachment of magnetically labelled or fluorescent antibodies on to specific fetal cell surface markers. The resulting sample is unsuitable for traditional cytogenetic analysis because it is still highly contaminated with maternal cells. However, with the use of chromosome- specific DNA probes and fluorescent in situ hybridization (FISH), it is possible to suspect fetal trisomy by the presence oFthree-signal nuclei in some of the cells of the maternal blood enriched for fetal cells.
On the basis of currently available technology, examination of fetal cells from maternal peripheral blood is more likely to find an application as a method for assessment of risk, rather than the non-invasive prenatal diagnosis of chromosomal defects. The sensitivity of this method is comparable to serum screening. However, unlike serum biochemistry testing, which is relatively easy to apply for mass population screening, analysis of fetal cells from maternal blood is both labor intensive and requires highly skilled operators. The extent to which the techniques for enrichment of fetal cells could be improved, to achieve a higher yield of the necessary cells, as well as become automated, to allow simultaneous analysis of a large number of samples, remains to be seen.
Recent interest has focused on the presence of cell-free fetal DNA in maternal plasma and the ability to quantify the concentration of male fetal DNA in pregnancies with male fetuses using real-time quantitative PCR. There is contradictory evidence concerning the concentration of cell-free fetal DNA in trisomy 21 pregnancies with some studies reporting that the levels are increased and in others there was no significant difference from chromosomally normal pregnancies. The extent to which cell-free fetal DNA will become another maternal serum marker in screening for trisomy 21 remains to be seen.
Still in India NIPT is NOT well-accepted method of the screening because of the prohibitive cost and NO LAB support in India at present! These samples are outsourced to other country like China or USA!
At present, the cost effectiveness is questionable for the developing or poor countries like India.
As almost about 80 % of the fetuses are screened by NT (ultrasound markers e.g. Nasal bone, ductus venosus )and can be classified as LOW risk.
About 5% are classified as HIGH risk on the basis of NT (ultrasound markers e.g. Nasal bone, ductus venosus) , and offered invasive testing for either for NIPT or chromosomal study after CVS.
Rest of about 15% who are in to intermediate risk, we can take help of biochemical markers to put them in to HIGH or LOW risk. If still couple wants to confirm the diagnosis, couple may be offered the prenatal invasive diagnosis or NIPT.
The advantages of first-trimester screening extend beyond the observed higher detection rates. The main advantage for the majority of women is the earlier reassurance provided to those with ‘low-risk’ results.
Those who are screen positive can have a CVS and diagnosis by the 14th week. Complication rates are also lower at earlier gestations for termination of pregnancy in case of fetal chromosomal anomalies.
A range of abnormalities other than aneuploidy has been associated with increased nuchal translucency. These include cardiac anomalies, diaphragmatic hernia, skeletal dysplasia, and abnormal lymphatic drainage associated with neuromuscular disorders and many more.
Nicolaides KH et al 23 mentioned that almost 43.6% of fetal anomalies were detected at 11-13+6 weeks. The 11- 13+6 weeks scan detected all cases of acrania,
alobarholoprosencephaly,exomphalos, gastroschisis, megacystis and body stalk anomaly, 77% of absent hand or foot,50% of diaphragmatic hernia, 50% of lethal skeletal dysplasias, 60% of polydactyly, 34% ofmajor cardiac defects, 5% of facial clefts and 14% of open spina bifida, but none of agenesis ofthe corpus callosum, cerebellar or vermian hypoplasia, echogenic lung lesions, bowelobstruction, most renal defects or talipes. Nuchal translucency (NT) was above the 95thpercentile in 34% of fetuses with major cardiac defects.
Therefore, identification of an increased nuchal translucency measurement should prompt not only a diagnostic test for aneuploidy, but also a thorough anatomic survey for structural anomalies and a detailed fetal echocardiogram in the case of a normal karyotype.
A significant number of anomalies can be picked up at this gestational age. With the increasing use of higher resolution machines a detailed anatomical survey can be done at the 11- 13 week scan.
In our country the baseline awareness of Down syndrome is low and that for the screening test even less. Counseling needs to be one on one, ensuring that the information is understood correctly.
Once the screening is done, the result is again explained. If screen positive, the details of invasive testing are discussed and appointment for the same scheduled.
Important table to understand:
During the last 30 years, extensive research has aimed at developing a non-invasive method for prenatal diagnosis based on the isolation and examination of fetal cells found in the maternal circulation. About 1 in 103–107 nucleated cells in maternal blood are fetal. The proportion of fetal cells can be enriched to about 1 in 10–100 by techniques such as magnetic cell sorting (MACS) or fluorescence activated cell sorting (FACS) after attachment of magnetically labelled or fluorescent antibodies on to specific fetal cell surface markers. The resulting sample is unsuitable for traditional cytogenetic analysis because it is still highly contaminated with maternal cells. However, with the use of chromosome- specific DNA probes and fluorescent in situ hybridization (FISH), it is possible to suspect fetal trisomy by the presence oFthree-signal nuclei in some of the cells of the maternal blood enriched for fetal cells.
On the basis of currently available technology, examination of fetal cells from maternal peripheral blood is more likely to find an application as a method for assessment of risk, rather than the non-invasive prenatal diagnosis of chromosomal defects. The sensitivity of this method is comparable to serum screening. However, unlike serum biochemistry testing, which is relatively easy to apply for mass population screening, analysis of fetal cells from maternal blood is both labor intensive and requires highly skilled operators. The extent to which the techniques for enrichment of fetal cells could be improved, to achieve a higher yield of the necessary cells, as well as become automated, to allow simultaneous analysis of a large number of samples, remains to be seen.
Recent interest has focused on the presence of cell-free fetal DNA in maternal plasma and the ability to quantify the concentration of male fetal DNA in pregnancies with male fetuses using real-time quantitative PCR. There is contradictory evidence concerning the concentration of cell-free fetal DNA in trisomy 21 pregnancies with some studies reporting that the levels are increased and in others there was no significant difference from chromosomally normal pregnancies. The extent to which cell-free fetal DNA will become another maternal serum marker in screening for trisomy 21 remains to be seen.
Still in India NIPT is NOT well-accepted method of the screening because of the prohibitive cost and NO LAB support in India at present! These samples are outsourced to other country like China or USA!
In the developing countries, I think 90 % detection rate is good enough and also cost effective, doing screening by ONLY Ultrasonography with appropriate training. As serum biochemistry is more costly than 11-13 weeks scan in India!As Ibelieve than screening tools should be cheap and available across the country. Very few, accredited with FMF, labs at present are working in India. Unfortunately, Indian subcontinent is very hot. Serum markers like PAPP-A and b-hCG are very sensitive to hot climate. This situation makes it difficult to send the blood samples to different centers as it is going to produce incorrect result and HIGH false positive tests leading to unwarranted fetal interventions!
